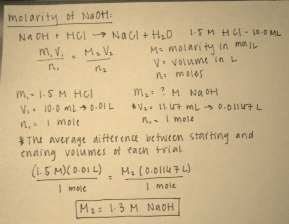Purpose: The purpose of this experiment was to determine the concentration of NaOH (sodium hydroxide) solution by preforming a titration using HCl solution of known concentration.
Procedure:
1. A buret is prepared with the titrant solution of HCl, which is the solution of known concentration of 1.5M. The initial volume is recorded.
2. An Erlenmeyer flask is prepared with NaOH, the solution with unknown concentration. The volume of this solution is recorded.
3. Two drops of the chosen indicator (in this case phenolpthalein) is added to the flask.
4. The stopcock is opened, releasing a stream of titrant into the flask. The indicator will change color when the titrant first makes contact, but after stirring it will return to clear.
5. Step 4 is repeated, releasing less and less titrant as the endpoint is approached. A pipet can be used to add small drops of titrant to the flask.
6. For the indicator phenolpthalein, the endpoint is marked by a change in color to light pink that lasts for at least 30 seconds before turning clear again.
7. Once the liquid maintains this light pink color for at least 30 seconds, the volume of titrant solution is recorded and the difference in volume calculated. Using these measurements and the known concentration of the titrant solution, the concentration of the other solution can be calculated.
2. An Erlenmeyer flask is prepared with NaOH, the solution with unknown concentration. The volume of this solution is recorded.
3. Two drops of the chosen indicator (in this case phenolpthalein) is added to the flask.
4. The stopcock is opened, releasing a stream of titrant into the flask. The indicator will change color when the titrant first makes contact, but after stirring it will return to clear.
5. Step 4 is repeated, releasing less and less titrant as the endpoint is approached. A pipet can be used to add small drops of titrant to the flask.
6. For the indicator phenolpthalein, the endpoint is marked by a change in color to light pink that lasts for at least 30 seconds before turning clear again.
7. Once the liquid maintains this light pink color for at least 30 seconds, the volume of titrant solution is recorded and the difference in volume calculated. Using these measurements and the known concentration of the titrant solution, the concentration of the other solution can be calculated.
Data Table:
|
Trial #
Trial 1 Trial 2 Trial 3 |
Starting Volume
6.8 mL 19.8 mL 31.2 mL |
Ending Volume
19.5 mL 31.0 mL 42.3 mL |
Difference
12.7 mL 11.2 mL 11.1 mL |
Calculations:
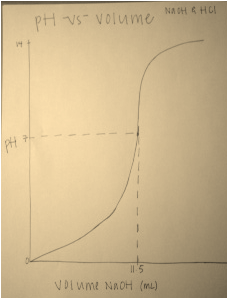
Graph: The dot with identifying lines is the equivalence point for this titration.
Conclusion
After completing the titration, it was calculated from the measurements gathered during the experiment that the molarity of sodium hydroxide is 1.3 moles per liter. This was determined by measuring the changes in volume before and after each trial. The average volume was then divided by the standard molarity and volume of hydrochloric acid. The result is the molarity of sodium hydroxide. This value of 1.3 moles per liter is reasonable, seing as the molarity of hydrochloric acid in this experiment was 1.5 molar. The titration method is a very useful and efficient way to determine the molarity of an unknown solution (in this case sodium hydroxide) from one that has known concentration. The key is to use the appropriate indicator, which is one that will change color at the end of the reaction. This is the key to a successful titration, and determining the perfect indicator can be done by using major ideas of pH and equilibrium.
Discussion of Theory
A titration is a seemingly simple and common experiment, but in actuality can be almost impossible to preform correctly. A titration reaction is reliant on the neutralization of an acid and base when mixed in solution. In this titration, the acid used was hydrochloric acid with a known concentration (known as the titrant), and the base was sodium hydroxide with an unknown concentration. In addition to the hydrochloric acid solution, the indicator phenolpthalein was added. This indicator was chosen because its pH range is that of the equivalence point of the reaction. The indicator is added to visually demonstrate the pH range of the equivalence point. The equivalence point is when the reaction between the titrant and other solution is complete. At this point the concentration of hydrogen ions equals that of hydroxide ions. This is not to be confused with the end point, which is when the indicator changes color. In a good titration, the equivalence point and the end point are the same, giving a fairly accurate pH range for this point in the reaction. Most indicators are weak acids that shift from acid to conjugate base forms during the reaction. A proton is lost or gained during the transition from acid to conjugate base, marking the endpoint. The release of this proton causes a change in the pH of the solution (since pH measures the negative log of the hydrogen ion concentration) that causes the color change. The best indicator for a particular titration is chosen based on the equivalence point of the reaction being preformed. Most common indicators have been experimented with for many years, so the range of their endpoints is standardized. Scientists use this and the equivalence point of the reaction to choose the indicator whose end point range contains the equivalence point of the desired reaction. For example, in this titration the indicator phenolpthalein was used to show the equivalence point. In its weak acid form, this indicator appears clear; when it has transitioned into its ion form, it is very bright pink. At the endpoint, phenolpthalein turns faint pink, barely visible in solution. If the solution turns bright pink , too much base was added and the pH has exceeded that of the equivalence point due to an increased number of hydrogen ions from the large number of indicator ions. The solution is clear prior to the equivalence point, because there is not a large concentration of indicator or hydrogen ions to change the pH. When the drops of base first make contact with the acid solution, it turns bright pink and fades back to clear with stirring. The equivalence point is reached when the faint pink color is sustained for a minimum of 30 seconds before the solution returns to clear. Adding additional hydrogen ions shifts equilibrium left, according to LeChatelier's principle, causing the color to appear clear. Adding hydroxide ions to the solution neutralizes the hydrogen ions, shifting equilibrium back to the right, resulting in the color change to pink This color change occurs at around a pH of 9-10.
LeChatelier's principle describing the equilibrium shifts of hydrogen and hydroxide ions is the heart of the chemistry behind a titration. The shifts in concentration of hydrogen and hydroxide can be explained by this principle, and are the reason this method works for determining the concentration for an unknown substance. As the number of hydrogen ions is increased with the addition of strong acid, this shifts equilibrium left. But these hydrogen ions are neutralized by the subsequent increase in hydroxide ions from the strong base in the reverse reaction. The reaction for a strong acid/base titration at the equivalence point is as follows: H+ + OH- yields H2O. Since the concentrations of hydrogen and hydroxide ions are equal, they will react to form water with a pH of 7 (neutral). Water is the only thing (besides a neutral salt) that remains in the solution, because the hydrogen and hydroxide ions neutralize each other. This is why the pH of a strong acid/base titration at the equivalence point is always 7.
Acid/base titrations are very useful procedures for determining the concentration of a solution. They can be preformed with combinations of strong and weak acids and bases, with variations to the equivalence points due to the concentrations of hydrogen and hydroxide at varying points of the reaction.
LeChatelier's principle describing the equilibrium shifts of hydrogen and hydroxide ions is the heart of the chemistry behind a titration. The shifts in concentration of hydrogen and hydroxide can be explained by this principle, and are the reason this method works for determining the concentration for an unknown substance. As the number of hydrogen ions is increased with the addition of strong acid, this shifts equilibrium left. But these hydrogen ions are neutralized by the subsequent increase in hydroxide ions from the strong base in the reverse reaction. The reaction for a strong acid/base titration at the equivalence point is as follows: H+ + OH- yields H2O. Since the concentrations of hydrogen and hydroxide ions are equal, they will react to form water with a pH of 7 (neutral). Water is the only thing (besides a neutral salt) that remains in the solution, because the hydrogen and hydroxide ions neutralize each other. This is why the pH of a strong acid/base titration at the equivalence point is always 7.
Acid/base titrations are very useful procedures for determining the concentration of a solution. They can be preformed with combinations of strong and weak acids and bases, with variations to the equivalence points due to the concentrations of hydrogen and hydroxide at varying points of the reaction.
Questions
1. The purpose of doing a titration is to calculate the concentration of a solution in which the concentration is unknown. This is done by titrating the solution of unknown concentration with a solution with known concentration.
2. An indicator is a weak acid or base that is used in chemical reactions to visually determine when the reaction has transitioned past the equivalence point, or the point in which the reaction between the titrant and the substance of unknown concentration is complete. The indicator will change color as soon as the pH changes as a result of this reaction. Indicators work by way of their own chemical reaction. Since most indicators are weak acids, during a reaction these acids form their conjugate base pairs. The color change occurs when the proton from the weak acid is donated to its product. It is this donation that causes a change in pH that triggers the color change.
3. An appropriate indicator is chosen for a reaction based on the equivalence point of the reaction. The best indicator will change color at the pH of the equivalence point of the reaction. Determining the equivalence point of a reaction is the purpose of adding an indicator, so the one chosen should change color around the expected pH of the equivalence point of the reaction.
4. The equivalence point is the point at which the indicator changes color. The endpoint of a reaction is when the reaction is complete. These points during a reaction are similar but not the same. In a good titration, the equivalence point should be the same as the end point. This can be achieved by using an indicator that changes color (marking the equivalence point) at the same time that the reaction ends (the endpoint).
5. SEE BELOW FOR GRAPHS AND EXPLANATION.
6. SEE BELOW FOR GRAPHS AND EXPLANATION.
7. A buffered solution is one that resists a change in pH due to the addition of protons (H+) or hydroxide (OH-) ions. These ions enter the solution as a result of the conjugate salt to the weak acid or base. An example of a buffer solution is hydrofluoric acid and sodium floride. The acid and its conjugate are in equilibrium, and according to LeChatilier's Principle, equilibrium will shift to counteract any changes in this balance. If an acid is added, this will increase the concentration of H+ ions, shifting equilibrium left to favor the reactants. But then the reverse reaction becomes unbalanced and shifts back right favoring the products of the forward reaction. This means the concentration of hydrogen ions remains nearly the same, and as a result, the pH does not change any measurable amount.
8. a) H: distilled water will decrease the concentration of HCl, so less NaOH will be used than necessary, increasing the molarity.
b) L: this would make the concentration of HCl is higher than expected, so more NaOH will be used, decreasing the molarity.
c) N: adding more indicator does not affect when the equivalence point is reached, so it will not affect the calculations of molarity of NaOH.
d) N: the air bubble did not change the volume used during the titration, which is the most important value in the calculations.
e) L: this would falsely increase the difference in starting and ending volume, making the volume used appear larger, diluting the molarity of NaOH.
f) L: same reasoning as (e).
g) L: the solution will never change color so way too much NaOH will be used and the equivalence point missed, so the artificially large volume of NaOH used will decrease its molarity.
2. An indicator is a weak acid or base that is used in chemical reactions to visually determine when the reaction has transitioned past the equivalence point, or the point in which the reaction between the titrant and the substance of unknown concentration is complete. The indicator will change color as soon as the pH changes as a result of this reaction. Indicators work by way of their own chemical reaction. Since most indicators are weak acids, during a reaction these acids form their conjugate base pairs. The color change occurs when the proton from the weak acid is donated to its product. It is this donation that causes a change in pH that triggers the color change.
3. An appropriate indicator is chosen for a reaction based on the equivalence point of the reaction. The best indicator will change color at the pH of the equivalence point of the reaction. Determining the equivalence point of a reaction is the purpose of adding an indicator, so the one chosen should change color around the expected pH of the equivalence point of the reaction.
4. The equivalence point is the point at which the indicator changes color. The endpoint of a reaction is when the reaction is complete. These points during a reaction are similar but not the same. In a good titration, the equivalence point should be the same as the end point. This can be achieved by using an indicator that changes color (marking the equivalence point) at the same time that the reaction ends (the endpoint).
5. SEE BELOW FOR GRAPHS AND EXPLANATION.
6. SEE BELOW FOR GRAPHS AND EXPLANATION.
7. A buffered solution is one that resists a change in pH due to the addition of protons (H+) or hydroxide (OH-) ions. These ions enter the solution as a result of the conjugate salt to the weak acid or base. An example of a buffer solution is hydrofluoric acid and sodium floride. The acid and its conjugate are in equilibrium, and according to LeChatilier's Principle, equilibrium will shift to counteract any changes in this balance. If an acid is added, this will increase the concentration of H+ ions, shifting equilibrium left to favor the reactants. But then the reverse reaction becomes unbalanced and shifts back right favoring the products of the forward reaction. This means the concentration of hydrogen ions remains nearly the same, and as a result, the pH does not change any measurable amount.
8. a) H: distilled water will decrease the concentration of HCl, so less NaOH will be used than necessary, increasing the molarity.
b) L: this would make the concentration of HCl is higher than expected, so more NaOH will be used, decreasing the molarity.
c) N: adding more indicator does not affect when the equivalence point is reached, so it will not affect the calculations of molarity of NaOH.
d) N: the air bubble did not change the volume used during the titration, which is the most important value in the calculations.
e) L: this would falsely increase the difference in starting and ending volume, making the volume used appear larger, diluting the molarity of NaOH.
f) L: same reasoning as (e).
g) L: the solution will never change color so way too much NaOH will be used and the equivalence point missed, so the artificially large volume of NaOH used will decrease its molarity.
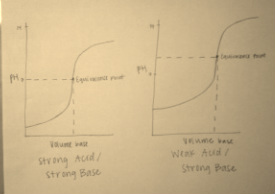
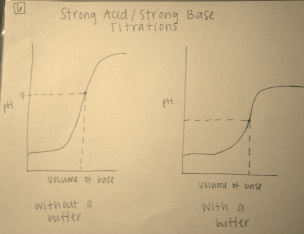
Questions 5 and 6:
5. Above are the graphs of a strong base/strong acid and strong base/weak acid titrations. As shown in the graphs, the starting pH in the strong acid titration is lower than that of the weak acid titration. This is because strong acids dissociate completely in solution, so they release a larger amount of hydrogen ions, thus decreasing the pH of the solution. Also, the equivalence point for the strong acid titration is seven, while the equivalence point for the weak acid is at a pH above seven. This is because the salt produced does not undergo any hydrolysis reactions. Both titrations follow a similar logarithmic function, but vary slightly due to the differing compositions of weak and strong acids.
6. The graphs above compare the titrations of a strong acid and strong base with and without the addition of a buffer. A buffered solution is one that resists any changes in pH. The addition of a buffer causes the increase in pH associated with the progression of the reaction to slow, and require larger volumes of strong base to be added to the strong acid to reach the equivalence point. Since the purpose of a buffer is to resist changes in pH, it makes sense that a graph of this type of titration would produce a graph that slopes more gently than a titration without a use of a buffer. This resistance to changes in pH results in a lower equivalence point for the titration with a buffer than the same without a buffer. Non-buffered solutions are able to reach higher pH levels at their equivalence points due to the lack of restriction of the salts in the buffered solutions.