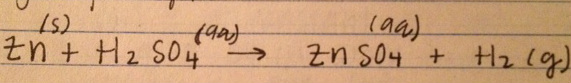Purpose:
The purpose of this experiment is to determine, through a series of various reactions, the concluding mass from a standard amount of copper, determining how copper is affected through different types of reactions, and how scientific laws hold during actual reactions.
Quantitative Data:
Initial Mass of Copper: 2.09 grams
Mass of Evaporating Dish: 52.28 grams
Mass of Evaporating Dish and Copper: 59.22 grams
Mass of Recorded Copper: 6.94 grams
Qualitative Data: reactions and observations from data table
Copper and Nitric acid: gas evolved, color change to blue, extremely hot.
Copper nitrate and Sodium hydroxide: blue precipitate formed, gas evolved
Copper (II) oxide and Sulfuric acid: color change to blue, became aqueous and transparent.
Copper sulfate and Zinc: gas evolved, brown precipitate pieces formed
Zinc and Sulfuric acid: very hot, gas evolved
Net Ionic Equations
Calculations
Conclusion
Based on the data gathered from the reactions, the final mass of copper is 6.94 grams, much larger than the starting mass of 2.09 grams. The percent yield, over 300%, reflects many sources of error throughout the various reactions.
Discussion of Theory
Throughout this laboratory experiment, a variety of reactions were completed, including single replacement, double replacement, and decomposition. A single replacement reaction, a type of oxidation reduction reaction, occurs when one free element replaces another element in a compound. During these reactions, cations (positively charged particles) replace other cations, and anions (negatively charged particles) replace other anions. For a single replacement reaction to occur, the metal or halogen replacing the other element must be higher on the Activity Series list. As one moves down this list, the elements become less and less reactive. To replace another element, the metal or halogen must be more reactive than the one it is trying to replace. If this is not the case, the reaction is said not to occur. In the reaction between copper and Nitric acid, copper replaced hydrogen, forming copper nitrate and hydrogen gas. This reaction theoretically occurred because copper is higher on the Activity series than hydrogen, so it was able to replace it during this reaction. A second type of reaction that occurred often during this experiment was double replacement. During this type of reaction, two elements with negative charges from two different compound "switch places" with each other, forming two new compounds on the product side. The negative elements move because they are more reactive than their positive counterparts. When a double replacement reaction occurs, one must look to the products and their states to determine if the reaction did indeed occur. If both products are aqueous, then the reaction did not occur. This is because the reactants start off aqueous, and if both products are aqueous, nothing changed about the states of the elements. The final reaction that occurred was a decomposition reaction of copper hydroxide. In this type of reaction, a compound breaks down into the individual elements that comprise the compound. However, the decomposition of hydroxides and metals is unique, and in this reaction produced copper (II) oxide and water. Throughout each of these reactions, the same sample of copper was manipulated, producing different chemicals and compounds, while, in theory, retaining its original mass.
As all of these reactions occurred, they obeyed the Law of Conservation of Mass. This law states that matter cannot be created nor destroyed during a chemical reaction. Matter cannot simply disappear or appear during a reaction; it can only change state, become a compound, or a free element. So based on this law, the final mass of copper should equal the starting mass, since copper is not created nor destroyed during any of the reactions. For example, when copper was mixed with Nitric acid, the same amount of copper was present at the end of the reaction, but in the form of copper nitrate. Matter can be rearranged during chemical reactions, like during a single or double replacement reaction described above, but the same amount should be still be present. As each of the reactions in this lab occurred, the mass of copper should have been equal to the initial amount of around two grams. Throughout this lab, the same sample of copper formed various different compounds, from copper hydroxide to copper (II) oxide. During all of these reactions, the mass of copper remained constant, for the Law of Conservation of Mass states it so. Stoichiometry can be used to illustrate how the mass remains constant during the experiment. If the mass of the copper was calculated during one of the many experiments, the results would yeild the same as the starting amount, proving the Law of Conservation of Mass. This law is also exemplified by the mole to mole ratio, which holds that the number of moles of the reatants is a ratio with those of the products, based on their coefficients. This ratio supports the Law of Conservation of Mass, because the same amount of moles of copper was present at the end of the reaction, just in different compounds. After completing the calculations to determine the final mass and moles of copper, it was determined that there was more than three times the amount of copper present than in the commencement of the experiment. This is due to a myriad of sources of error, which are discussed below. If this lab were completed without any sources of error, the Law of Conservation of Mass would prove correct, and the final mass of copper would be identical to the starting mass. This is very unlikely occur, for human and systematic errors prevent the amounts to be truly equal, but very close.
As all of these reactions occurred, they obeyed the Law of Conservation of Mass. This law states that matter cannot be created nor destroyed during a chemical reaction. Matter cannot simply disappear or appear during a reaction; it can only change state, become a compound, or a free element. So based on this law, the final mass of copper should equal the starting mass, since copper is not created nor destroyed during any of the reactions. For example, when copper was mixed with Nitric acid, the same amount of copper was present at the end of the reaction, but in the form of copper nitrate. Matter can be rearranged during chemical reactions, like during a single or double replacement reaction described above, but the same amount should be still be present. As each of the reactions in this lab occurred, the mass of copper should have been equal to the initial amount of around two grams. Throughout this lab, the same sample of copper formed various different compounds, from copper hydroxide to copper (II) oxide. During all of these reactions, the mass of copper remained constant, for the Law of Conservation of Mass states it so. Stoichiometry can be used to illustrate how the mass remains constant during the experiment. If the mass of the copper was calculated during one of the many experiments, the results would yeild the same as the starting amount, proving the Law of Conservation of Mass. This law is also exemplified by the mole to mole ratio, which holds that the number of moles of the reatants is a ratio with those of the products, based on their coefficients. This ratio supports the Law of Conservation of Mass, because the same amount of moles of copper was present at the end of the reaction, just in different compounds. After completing the calculations to determine the final mass and moles of copper, it was determined that there was more than three times the amount of copper present than in the commencement of the experiment. This is due to a myriad of sources of error, which are discussed below. If this lab were completed without any sources of error, the Law of Conservation of Mass would prove correct, and the final mass of copper would be identical to the starting mass. This is very unlikely occur, for human and systematic errors prevent the amounts to be truly equal, but very close.
Sources of Error
This lab had multiple steps and reactions completed, and therefore causing many sources of error. When the mixture of copper and sodium hydroxide was heated, it formed a large bubble that burst, removing copper from the sample. This explosion occurred because of a faulty hot plate, a systematic source of error. Also, when the various mixtures were stirred with a glass stirring rod, some copper remained on the rod, decreasing the amount of copper in the yield Furthermore, the stir rod was used incorrectly, further removing copper from the system. During one of the reactions, the copper mixture was decanted to remove excess water. During this process, some copper was left in the beaker. To remove this excess copper, water was piped into the beaker. This technique added additional water into the system, as well as removing copper along with the targeted water. On the other hand, when zinc was added and then removed during a reaction, some zinc remained in the copper mixture increasing the mass of the copper. The final mass of copper was elevated from its starting amount due to zinc remaining in the evaporating dish and other elements added throughout the experiment. It was expected that the final mass would be lower than the original due to the explosion that occurred early in the experiment; but excess zinc in the final measurement was to blame for the abnormally large data sample.
Questions
1. The product of the reaction between copper and Nitric acid in Step 2 was placed on ice because during the exothermic reaction, large quantities of heat were released. The product was placed in an ice bath to lower the temperature before beginning the later reactions, so the results recorded would be more accurate.
2. In Step 4, a double replacement reaction occurred between Copper nitrate and Sodium hydroxide, forming Copper hydroxide and Sodium nitrate. In Step 7, a double replacement reaction took place between Copper (II) oxide and Sulfuric acid, forming Copper sulfate and water. In Step 9, a single replacement reaction occurred between Zinc and Sulfuric acid, producing Zinc sulfate and Hydrogen gas.
3. The balanced reaction between Zinc and Sulfuric acid is below:
2. In Step 4, a double replacement reaction occurred between Copper nitrate and Sodium hydroxide, forming Copper hydroxide and Sodium nitrate. In Step 7, a double replacement reaction took place between Copper (II) oxide and Sulfuric acid, forming Copper sulfate and water. In Step 9, a single replacement reaction occurred between Zinc and Sulfuric acid, producing Zinc sulfate and Hydrogen gas.
3. The balanced reaction between Zinc and Sulfuric acid is below:
Some problems that could arise form an incomplete reaction of Zinc and Sulfuric acid are related to the final mass of copper. If this crucial reaction is not completed thoroughly, there will still be Zinc remaining in the solid copper when weighed in the evaporating dish. This will make the mass recorded higher than it actually is, accounting for a critical source of error for the experiment.
4. When the Copper (II) oxide was washed, hydroxide, sodium, and nitrate ions were removed from the reaction between copper nitrate and sodium hydroxide. These ions were spectator ions in the reaction, and while they are omitted from the net ionic equation, they are still present in the solution. Therefore, it is necessary to wash the Copper (II) oxide to remove these ions before proceeding to the next reaction.
5. After the Sulfuric acid was poured into the beaker, the copper was aqueous. This was seen through the lack of a precipitate formed and the colored, transparent solution in the beaker.
6. When the precipitated copper was washed, Zinc and Sulfate ions were removed. These ions formed during the reaction with Copper sulfate, which yielded Zinc sulfate and solid copper.
5. After the Sulfuric acid was poured into the beaker, the copper was aqueous. This was seen through the lack of a precipitate formed and the colored, transparent solution in the beaker.
6. When the precipitated copper was washed, Zinc and Sulfate ions were removed. These ions formed during the reaction with Copper sulfate, which yielded Zinc sulfate and solid copper.