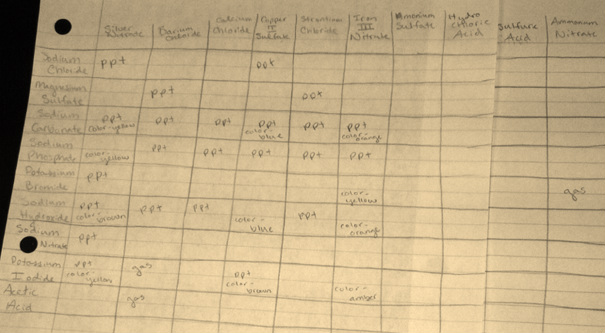
Data Table
Conclusion
It can be concluded from observing the reactions and interpreting its data, some reactions produce observable chemical changes when they occur, and these changes can be predicted by using solubility rules.
Discussion of Theory
There are many observable signs that a chemical reaction has taken place, including the formation of a precipitate, a color change, and the formation of a gas, seen through bubbles. In addition, the written reaction, once balanced, indicates the status of the reaction through the rules of solubility. The solubility rules identify which reactants, when bonded together, form precipitates. If both of the products formed are aqueous, or dissolved in water, the reaction did not occur. In this lab, various compounds, acids and bases were reacted and the outcomes observed and recorded. From these results, the general rules for solubility were determined. For example, in every reaction where a nitrate was bonded to another element on the product side, it did not form a solid precipitate. Therefore, it can be assumed that all nitrates are soluble, no matter what is bonded to them.
From the balanced chemical reaction, the net ionic equation can be derived. The net ionic equation only includes the ions that participated in the reaction. It is formed by breaking down the strong acids and bases into ions, and removing the spectator ions, or the ions that did not change from the reactant side to that of the products. The remaining ions comprise what is called the net ionic equation. This simplified reaction makes it much easier to identify what occurred during the reaction, by only including what ions reacted to form the products. The net ionic equations from various reactions in this experiment contained the ions that formed the precipitate, or any other indicator of the reaction. It is therefore very useful to determine the net ionic equation of a reaction, for it clearly demonstrates what ions truly caused the chemical reaction, while omitting those who did not contribute to the formation of the solid or gas.
From the balanced chemical reaction, the net ionic equation can be derived. The net ionic equation only includes the ions that participated in the reaction. It is formed by breaking down the strong acids and bases into ions, and removing the spectator ions, or the ions that did not change from the reactant side to that of the products. The remaining ions comprise what is called the net ionic equation. This simplified reaction makes it much easier to identify what occurred during the reaction, by only including what ions reacted to form the products. The net ionic equations from various reactions in this experiment contained the ions that formed the precipitate, or any other indicator of the reaction. It is therefore very useful to determine the net ionic equation of a reaction, for it clearly demonstrates what ions truly caused the chemical reaction, while omitting those who did not contribute to the formation of the solid or gas.
Sources of Error
There are many factors that caused error in this lab, by causing misinterpretation of the reactions, or lack of, taking place. The principle source of error was when observing the formation of a gas during a chemical reaction, the gas was from air forming in the pipet, not the chemical reaction. This was determined when the balanced reaction was written out and the solubility rules applied, two aqueous products were formed. A reaction did not occur when both products are aqueous, so there was error when the reaction was observed. Furthermore, some reactions yielded a gas as a product, but it was not detectable by the human eye during observation. The same is true for the formation of a precipitate. A second source of error in this experiment was the unequal amount of reactants being used during each experiment. Since a pipet was used, it is likely that the size of the drops of reactants were not all equal, creating error in the data later observed.
Anaylsis Questions
1. Based on the data, it can be determined that all Nitrates are soluble, no matter what molecule is bonded to them. Also, all Sulfates are soluble, except when bonded to Barium, Calcium, Strontium, and some other transition metals. All Phosphates are insoluble, with the exception of Group 1A elements and Ammonium. Hydroxides are also insoluble, other than when bonded to Barium, Group 1A elements and Strontium. All of these rules were demonstrated in the reactions that took place in the lab, and are further shown in the net ionic equations.
2. Net Ionic Equations from ten of the reactions recorded on the data table above:
3. Metathesis reactions are those in which cations and anions switch partners during the reaction. They do this because of the attraction between the positive and negative charges of cations and anions, respectively. There are three types of metathesis reactions, in which the products formed from aqueous reactants are different. In a precipitation reaction, the products formed are solids, called precipitates. A neutralization reaction forms neutral water molecules from an acid and a base. A gas formation reaction results in one product in the form of a gas. Examples of each type of metathesis reaction are listed below.
4. A molecule is soluble in water if the attraction between the polar water molecules is stronger than the forces bonding the molecules together. If the intermolecular attraction is weaker than the attraction to the water molecule, the water will completely envelope the molecule and cause it to dissociate, or break apart. If not, the molecule will stay together, making it insoluble in water. The process of water completely surrounding a molecule is called hydration. Hydration is the reason salt is not visible once placed in water; the salt molecules are completely surrounded by water molecules, making them to appear disappeared.
5. A double replacement reaction that produces two aqueous products is considered "no reaction" because the products did not change states, a definition of a reaction. Since both reactants are aqueous, having the products be aqueous means they did not change composition. Changing of composition of compounds is required for a reaction to take place, so two aqueous products do not constitute a reaction.
5. A double replacement reaction that produces two aqueous products is considered "no reaction" because the products did not change states, a definition of a reaction. Since both reactants are aqueous, having the products be aqueous means they did not change composition. Changing of composition of compounds is required for a reaction to take place, so two aqueous products do not constitute a reaction.