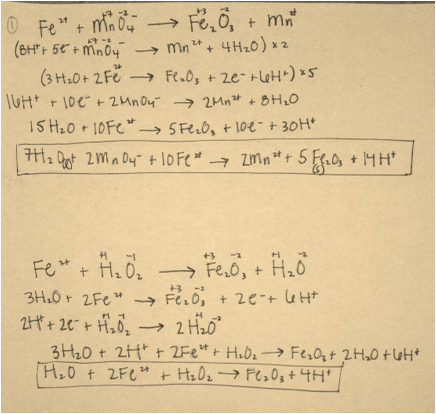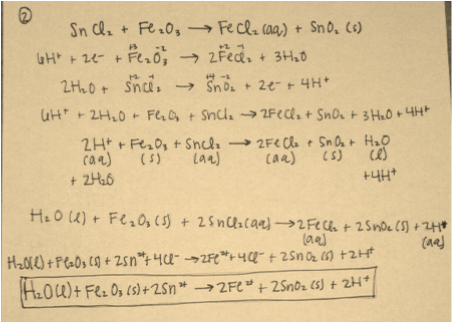Purpose: to observe the transfer of electrons during the processes of oxidation and reduction during each step of an oxidation-reduction reaction, and the effects this transfer has on the reaction.
Data Table:
|
Part
Part I Part II |
Observations
After the potassium thiocyanate solution was added, the solutions turned a transparent peach color. The KMno4- made the solution an opaque dark purple/red color. The H202 made the solution dark purple and opaque as well, but lighter than the previous solution. When stannous chloride was added, each solution became lighter red colored and much thinner. A precipitate formed in each solution, having the appearance of silver to brown flecks. It took approximately five seconds after stirring of the solution had ceased for the solution to return to a clear color. Stirring the solution caused it to turn a very deep blue once a few drops of methylene blue was added. |
Conclusion
After writing the oxidation-reduction reactions for those that occured during this experiment, the transfer of electrons between substances was clear. It is this transfer of electrons that classifies these reactions as oxidation-reduction reactions. In the reaction between iron and permanganate, iron was oxidized from 2+ to 3+ and manganese was reduced from +7 to +2. In the reaction between iron and hydrogen peroxide, iron was oxidized in the same manner as in the previous reaction, and oxygen was reduced from -1 to +1 in the hydrogen peroxide. Both these reactions show that iron generally behaves in the manner when in a oxidation-reduction reaction, revealing a pattern in the oxidation of iron and other elements. An oxidation-reduction reaction also occurred in the solution of methylene blue, potassium hydroxide and dextrose ( also known as glucose). Oxygen in the flask causes the oxidation of methylene blue once the flask is shaken, and glucose reduces methylene blue. This is a reversible chemical reaction that exhibits the properties of oxidaiton-reduction reactions as well as the principles of chemical equilibrium.
Discussion of Theory
Oxidation-reduction reactions are comprised of two reactions that occur in tandem; oxidation cannot occur without reduction and vice versa. Oxidation is the loss of electrons, resulting in a decrease in charge of the compound. Reduction, the opposide of oxidation, is the gain of electrons that causes the charge of the specie to increase. This increase and decrease in the number of electrons is also known as a change in the oxidation state of a compound. The oxidation state is the hypothetical charge of an atom if all the bonds between the atom and those of other elements were ionic. This means that the electrons are completely transfered from the atoms of one element to those of another; they are not shared as in a covalent bond. The charge is said to be hypothetical because no perfect ionic bond exists; the two atoms will always share a small portion of the "transfered" electron. The sharing is predominately on one side of the pair, making it seem like the electron was completely transfered. The oxidation states of elements are represented by their oxidation numbers, and it is these numbers that show whether the element was oxidized or reduced during the chemical reaction. There are general rules for assigning elements oxidation numbers that are valid in all oxidation-reduction reactions. For example, the oxidation number of an unbonded element (called a free element) is zero. In addition, certain ions tend to have the same oxidation numbers no matter what compound in which they are bonded. Examples are hydrogen (usually +1) and oxygen (most often -2). The oxidation numbers multiplied by the subscript of the elements must add to the overall charge of the compound. Once oxidation numbers are assigned to all elements in a reaction, it is clear which elements were oxidized and which were reduced. A decrease in the oxidation number means the charge decreased due to the addition of electrons (reduction). Conversely, an increase in the oxidation number signals a decrease in the number of electrons and thus a higher charge (oxidation). The processes of oxidation and reduction are often extracted from the complete reaction to analyze the individual transfers of electrons in each step. These are called half reactions, because the oxidation reaction cannot occur without the reduction reaction; they each make up half of the entire oxidation-reduction reaction. By isolating the oxidation and reduction reactions, it becomes much simpler to determine the number of electrons transfered and subsequently to balance the reaction.
The balancing of an oxidation-reduction reaction requires more steps than simply balancing the number of atoms on each side. The number of electrons transfered must be the same for each side of the reaction, once the half reactions are reunited in the full reaction. Also, the charge on each side must be equal. The method for balancing the charge is different based on the pH of the solution in which the reaction occurs. If the solution is acidic, meaning its pH is less than seven, hydrogen ions are used to balance the charge, and water molecules balance the number of hydrogen and oxygen atoms. If the reaction occurs in basic solution, where the pH is greater than 7, hydroxide ions balance charge and water molecules balance hydrogen and oxygen atoms. Using hydrogen and hydroxide ions for acidic and basic solutions, respectively, accurately represents the reactions occurring in the solutions. Oxidation-reduction reactions involve a transfer of electrons as the reaction progresses that is the cause of many phenomena seen in everyday life.
The balancing of an oxidation-reduction reaction requires more steps than simply balancing the number of atoms on each side. The number of electrons transfered must be the same for each side of the reaction, once the half reactions are reunited in the full reaction. Also, the charge on each side must be equal. The method for balancing the charge is different based on the pH of the solution in which the reaction occurs. If the solution is acidic, meaning its pH is less than seven, hydrogen ions are used to balance the charge, and water molecules balance the number of hydrogen and oxygen atoms. If the reaction occurs in basic solution, where the pH is greater than 7, hydroxide ions balance charge and water molecules balance hydrogen and oxygen atoms. Using hydrogen and hydroxide ions for acidic and basic solutions, respectively, accurately represents the reactions occurring in the solutions. Oxidation-reduction reactions involve a transfer of electrons as the reaction progresses that is the cause of many phenomena seen in everyday life.
Data Analysis Questions: The reactions for numbers 1 and 2 are in the pictures below.
1. Hydrogen peroxide has more oxidizing power than permanganate because it has a lower reduction potential value on the Reduction Potentials Table. This indicates that is hydrogen peroxide is more likely to be oxidized, which is the definition of oxidizing power. Since permanganate has a higher reduction potential, it is more likely to be reduced than oxidized, reducing its oxidizing power.
4. The colorless solution of methylene blue results from the reduction of methylene blue. The glucose reduces the methylene blue, meaning it is an oxidizing agent. When there is no more glucose in the solution, it will return to blue. The blue color results from the oxidation of methylene blue, and the powerful reducing agent glucoside. Glucoside forms from the reaction between glucose and hydroxide ions present from the potassium hydroxide. Shaking the flask containing the solution starts a reaction between oxygen dissolved in the solution and the reduced methylene blue. After standing for a few minutes, oxidized methylene blue reacts with glucoside forming reduced methylene blue.
5. The methylene blue no longer turns blue after many cycles and the reaction runs down because there is no more dissolved oxygen to oxidize the methylene blue. In theory, this experiment would be completed in a stoppered flask, so no oxygen could enter the closed system. After many trials, there would be no more dissolved oxygen to react with the reduced methylene blue to yield the oxidized form that has the blue color.
4. The colorless solution of methylene blue results from the reduction of methylene blue. The glucose reduces the methylene blue, meaning it is an oxidizing agent. When there is no more glucose in the solution, it will return to blue. The blue color results from the oxidation of methylene blue, and the powerful reducing agent glucoside. Glucoside forms from the reaction between glucose and hydroxide ions present from the potassium hydroxide. Shaking the flask containing the solution starts a reaction between oxygen dissolved in the solution and the reduced methylene blue. After standing for a few minutes, oxidized methylene blue reacts with glucoside forming reduced methylene blue.
5. The methylene blue no longer turns blue after many cycles and the reaction runs down because there is no more dissolved oxygen to oxidize the methylene blue. In theory, this experiment would be completed in a stoppered flask, so no oxygen could enter the closed system. After many trials, there would be no more dissolved oxygen to react with the reduced methylene blue to yield the oxidized form that has the blue color.